 To ensure that no center of mass motion occurs, the center atom (yellow ball) will also move. Symmetric Stretch Asymmetric Stretch Twistingįigure 3: Types of Vibrational Modes. A CH 2 group is used as an example to illustrate stretching and bending vibrations below. Four bending vibrations exist namely, wagging, twisting, rocking and scissoring. A change in the angle occurring between two bonds is known as a bending vibration. When there is a continuous change in the interatomic distance along the axis of the bond between two atoms, this process is known as a stretching vibration. This is not the case for large molecules due to several vibrational motions and interactions that will be experienced. These movements are easily defined for diatomic or triatomic molecules. 
Vibrational motions are defined by stretching and bending modes. This causes the atom not to be stationary and to fluctuate continuously. The dipole moment acts in the direction of the vector quantity. r i is the vector representing the position of i t h charge. q i is the magnitude of the i t h charge, and. Its a vector quantity, which means it has both magnitude and defined directions. The dipole moment of a molecule can be calculated by Equation 8.8.1: (8.8.1) i q i r i. The dipole moment of a molecule is therefore the vector sum of the dipole moments of the individual bonds in the molecule. The bond of a molecule experiences various types of vibrations and rotations. The bond dipole moment is the dipole moment of a single bond in a polyatomic molecule, which is distinct from the molecules overall dipole moment. The fifth term represents the interaction between the vibration and rotational interaction of the molecule. The second term represents anharmonicity and the fourth term represents centrifugal stretching. The first and third terms represent the harmonicity and rigid rotor behavior of a diatomic molecule such as HCl. Dipole moments occur in polar covalent bonds where electrons are shared unevenly between the two atoms. (a) There is a small difference in electronegativity between \ce molecule is bent, leading to a dipole moment.\] Dipole Moment: A dipole moment is defined as a measurement of the polarity of a molecule. 
The length of the arrow is proportional to the magnitude of the electronegativity difference between the two atoms.įigure 7.3.1. 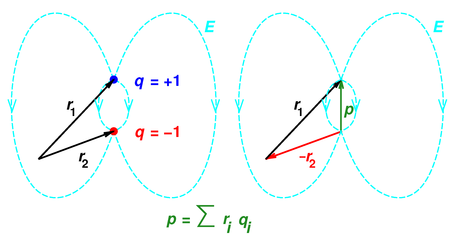
A small plus sign is drawn on the less electronegative end to indicate the partially positive end of the bond. Dipole vectors are shown as arrows pointing along the bond from the less electronegative atom toward the more electronegative atom. This bond moment can be represented as a vector, a quantity having both direction and magnitude ( Figure 7.3.1). The magnitude of a bond dipole moment is represented by the Greek letter mu ( µ) and is given by the formula shown below, where Q is the magnitude of the partial charges (determined by the electronegativity difference) and r is the distance between the charges: 
This separation of charge gives rise to a bond dipole moment. Assess the polarity of a molecule based on its bonding and structureĪs discussed previously, polar covalent bonds connect two atoms with differing electronegativities, leaving one atom with a partial positive charge (\delta+) and the other atom with a partial negative charge (\delta–), as the electrons are pulled toward the more electronegative atom. Electric dipole moment definition :- The dipole moment of an electric field is a vector whose magnitude is charge times the separation between two opposite. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
