|
1/20/2024 0 Comments Pf3 electron domain geometry 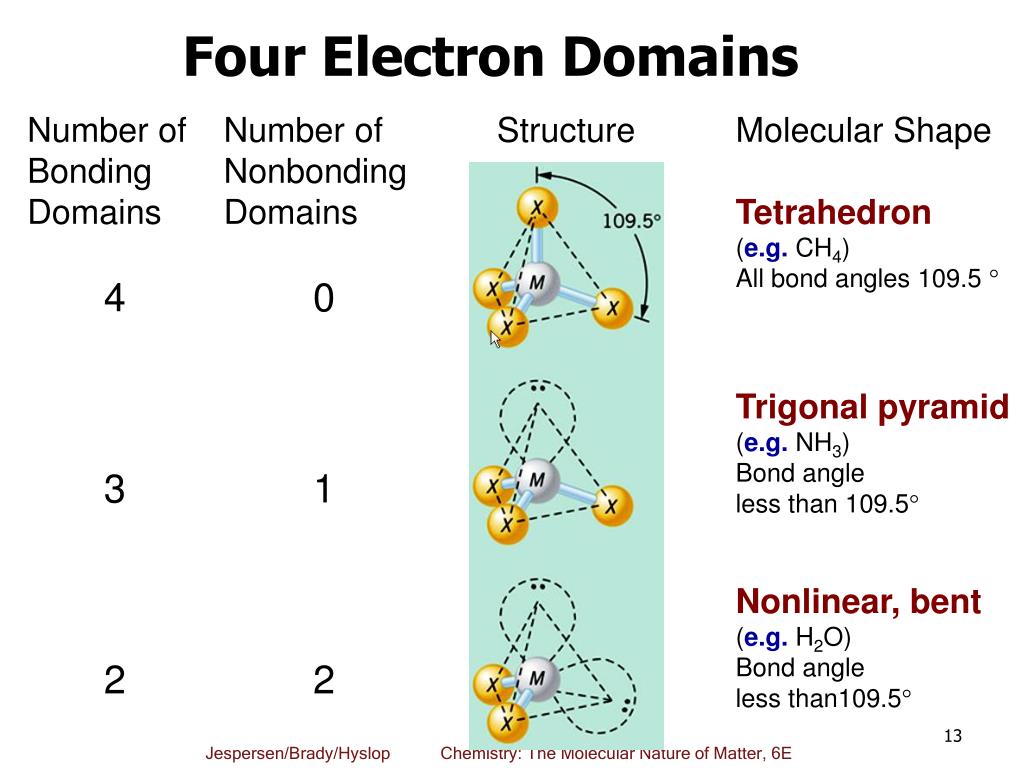
Here, in this case, the central electron does not have any octet as it has six particles. One thing to keep in mind while drawing Lewis structure is that the Octet Rule can be violated in these three situations but, we don’t need to think about it each time as it is rare and these exceptions will only occur when necessary.Įxception 1: If there is an odd number of valence electrons like 3,5,7, etc.Įxception 2: If there are very few valence electronsĮxception 3: If there are too many valence electrons But, as we know, there are no extra electrons. whether the molecule PF3 is polar or non-polar. Because the methane molecule has four electron domains (four hydrogen atoms and no lone pai. electron domain geometry, molecular geometry and bond angle/s. Then, add octets to the outer atom and extra electrons to the central atom. In this video we look at the electron geometry for Methane (CH4). As discussed, here there are 24 electrons. It’s like peripheral atoms all in one plane, as all three of them are similar with the 120° bond angles on each that makes them an equilateral. electron pair geometry of pf3 What Is The Electron Geometry Of Pf3. With the reference of Chemistry, ‘Trigonal Planar’ is a model with three atoms around one atom in the middle. domain geometry for a molecule with four electron pairs is tetrahedral. To draw a Lewis Structure, first of all, add electrons and draw the connectivities. The geometry of molecule of BF3 is ‘Trigonal Planar.’. If we check the formal charges for the Boron Trifluoride Lewis structure, we will find that they are zero even though Boron only had six valence electrons. It requires six valence electrons in its outer shell. Transcribed image text: The molecular geometry of the PF3 molecule is. Before completing the octets, don’t forget to determine how many valence electrons there in Boron Trifluoride and place them accordingly.īoron will be at the center of the structure because of being least electronegative. Lets count the areas around the phosphorus atom that contain at least two electrons. The hybridization of boron in BF 3 is sp 2. In the BF 3 Lewis dot structure, a total of 9 lone pairs and 3 bond pairs are present. The molecular geometry, on the other hand, is Trigonal Pyramidal. The electron geometry of BF 3 is also Trigonal planar, as its central atom, is surrounded by the 3 regions of electron density. 
This pair exerts repulsive forces on the bonding pairs of electrons. The electron geometry ('Electronic Domain Geometry') for PF3 is tetrahedral. The shape is distorted because of the lone pairs of electrons. It has a molecular geometry of trigonal pyramidal which also looks like a distorted tetrahedral structure. BF3 has a total of 24 valence electrons, which we have to set around the central atom. There are three single bonds and one lone pair of electrons in the NH3 molecule. Learn vocabulary, terms, and more with flashcards, games, and other study tools. According to VSEPR theory, if there are three electron domains in the valence shell of an atom, they will be arranged in a (n) geometry. To know about BF3 Lewis structure, we have to calculate the total number of valence electrons for the BF3 molecule. All of these will have bond angles of 120°. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
