If you have received all recommended doses of a COVID-19 vaccine that has been authorized or approved by the U.S. Learn more about developing COVID-19 vaccines. authorized or approved COVID-19 vaccines using data from the Vaccine Safety Datalink (VSD), Vaccine Adverse Event Reporting System (VAERS), and other safety monitoring systems. CDC and FDA continue to provide updated information on the safety of U.S. Hundreds of millions of people in the United States have safely received COVID-19 vaccines. Tracking Safety Using Vaccine Monitoring Systems – Like every other vaccine approved for use in the United States, COVID-19 vaccines continue to be monitored for safety and effectiveness.FDA has determined COVID-19 vaccines meet FDA’s standards and has granted those vaccines Emergency Use Authorizations (EUAs) or full FDA approval. Food and Drug Administration (FDA) reviews data from clinical trials. Authorization or Approval – Before vaccines are available to people, the U.S.The phases overlapped to speed up the process, but all phases were completed. Clinical Trials – All vaccines in the United States must go through three phases of clinical trials to ensure they are safe and effective.All vaccine development steps were taken to ensure COVID-19 vaccine safety and effectiveness, including: Protect children aged 6 months to 5 years from hospitalization caused by COVID-19Īlthough COVID-19 vaccines were developed quickly, research and development on vaccines like these has been underway for decades.Learn more about vaccination considerations and the safety and effectiveness of COVID-19 vaccines for people who are pregnant or breastfeeding. 
Protect babies younger than 6 months old from hospitalization caused by COVID-19.Prevent severe illness and death in people who are pregnant.People who get a COVID-19 infection during pregnancy are more likely to deliver a preterm (born earlier than 37 weeks of pregnancy) or stillborn infant and may also be more likely to have other pregnancy complications.ĬOVID-19 vaccination during pregnancy helps: In cases where you cannot get or choose not to get an mRNA vaccine, you may be offered the Novavax vaccine as your additional dose if you are 12 or older.Yes, COVID-19 vaccination is recommended for people who are pregnant, breastfeeding, or trying to get pregnant now, as well as people who might become pregnant in the future. The Pfizer/BioNTech vaccine is the recommended COVID-19 vaccine for an additional dose. People with a weak immune system are offered an additional dose for better protection. If you had the Novavax vaccine as your first round of COVID-19 vaccination, further doses of Novavax may be considered as boosters after a risk-benefit discussion with your vaccinator. If you are getting Novavax for a booster dose, you should get this 4 to 9 months after your last COVID-19 vaccine. This is only for people who cannot get or have chosen not to get an mRNA vaccine as your booster dose. If you had your first round of vaccination with Pfizer, Moderna or AstraZeneca, you may be offered the Novavax vaccine as a booster dose if you are 12 or older. You can get the Novavax vaccine as your second dose, 28 days after your first COVID-19 vaccine dose. This means you only had the first dose of either the Pfizer, Moderna or AstraZeneca vaccine. Not completed your first round of vaccination 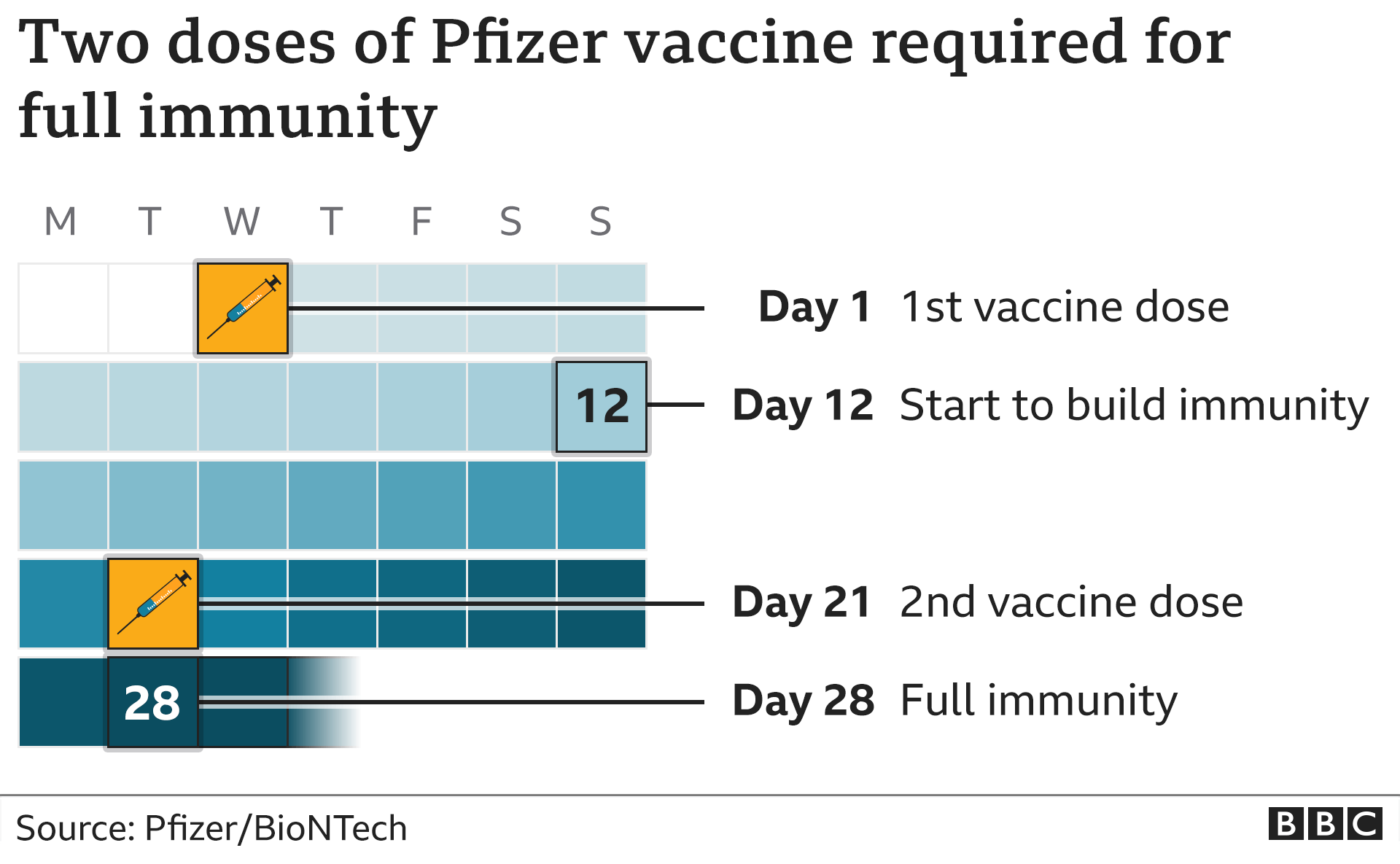
If you have never had a COVID-19 vaccine, you will need 2 doses of the Novavax vaccine, 21 days apart. The number of doses you need will depend on if you have been vaccinated against COVID-19 yet. Phone HSELive on 1800 700 700 to find out where you can get the Novavax vaccine. In this case, you may be offered the Novavax vaccine. For example, because of an allergy to one of the vaccine's ingredients. Some people may not be able to get this vaccine or have chosen not to get one. The Pfizer/BioNTech vaccine is the recommended COVID-19 vaccine for most people. 
Clinical trials are happening now to find this out. We do not know yet how long immunity will last after getting this vaccine. The Novavax COVID-19 vaccine is called Nuvaxovid. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
